Strategic Use of Bactrim in Vascular Patient Care
Disclaimer
This article is intended for informational purposes and does not constitute medical advice. Antibiotic therapy must be prescribed only after direct evaluation by a qualified healthcare professional. Inaccurate or self-directed antibiotic use may lead to complications or treatment failure.
Author: Charles D. Goff, MD, FACS
Why Bactrim Is Critical for Limb Infections in Vascular Patients
Bactrim (trimethoprim–sulfamethoxazole) remains an essential antimicrobial agent in managing limb infections among vascular patients, a population that faces substantially higher risks of tissue damage, impaired healing, and microbial colonization. Chronic ischemia, microvascular compromise, diabetes-related angiopathy, and neuropathy create an environment in which even minor wounds may progress to deep infections without timely intervention. Because these infections often involve Staphylococcus aureus, including MRSA, Bactrim offers a reliable oral option with strong activity against common pathogens.
In 2025, surgeons increasingly recognize that vascular patients often present with mixed soft-tissue infections arising from poor perfusion and chronic ulceration. These infections can escalate rapidly, leading to limb-threatening complications. Bactrim’s dual mechanism blocking sequential steps in bacterial folate synthesis provides potent activity against many of the organisms responsible for early ulcer infection and superficial tissue involvement. Its effectiveness, combined with good oral bioavailability, makes it especially suitable for outpatient care and early postoperative management.
The drug also fits well into modern antibiotic stewardship strategies because it avoids the broad-spectrum footprint of fluoroquinolones and carbapenems while still delivering meaningful coverage for resistant pathogens.
For clinicians seeking a balance between potency, safety, and preservation of future antimicrobial options, Bactrim remains one of the most strategically sound choices for limb infections in vascular patients.
Microbiology of Limb Infections in Vascular Compromise
Understanding the microbial landscape of vascular-limb infections is essential for selecting an appropriate antimicrobial regimen. Reduced tissue perfusion alters immune function and promotes colonization by organisms that thrive in stagnant, hypoxic environments. MRSA remains one of the most clinically significant pathogens in this population, particularly in patients with previous hospitalization, prior antibiotic exposure, or chronic ulceration.
Gram-negative organisms such as Enterobacteriaceae may be present in deeper or long-standing wounds, but early-stage infections typically remain staphylococcal in origin. The compromised microenvironment also encourages the formation of biofilms, which complicate treatment by reducing antibiotic penetration and delaying healing.
These factors collectively justify the use of Bactrim in early or superficial infections before anaerobic contamination or deep necrosis develops.
Recognizing this pattern allows surgeons to intervene earlier and reduce progression toward severe infection or limb-threatening complications.
When Bactrim Is the Preferred Agent in Vascular Limb Infections
Bactrim is particularly effective in vascular patients when the infection is superficial or moderate in depth and when MRSA is a concern. Chronic ischemic ulcers often begin with limited soft-tissue involvement, where the bacterial load is dominated by staphylococcal species rather than anaerobes. In such cases, Bactrim provides coverage well matched to the microbiology and helps prevent escalation to deep-tissue infection.
Clinical scenarios where Bactrim is especially appropriate include:
- Infected trophic ulcers without necrosis or foul odor, indicating limited anaerobic activity.
- Superficial MRSA-positive wounds, where oral therapy is desirable.
- Localized cellulitis in patients with chronic vascular disease who do not require hospitalization.
- Postprocedural infections after minor foot or leg interventions.
- Outpatient follow-up after initial debridement, where Bactrim can maintain coverage as the wound stabilizes.
These indications align with broader vascular-surgery infection protocols, including the antibiotic principles described in article Antibiotics in Vascular Surgery, where the management of wounds after bypass and other vascular procedures emphasizes early, targeted antimicrobial therapy to prevent graft contamination and limb deterioration.
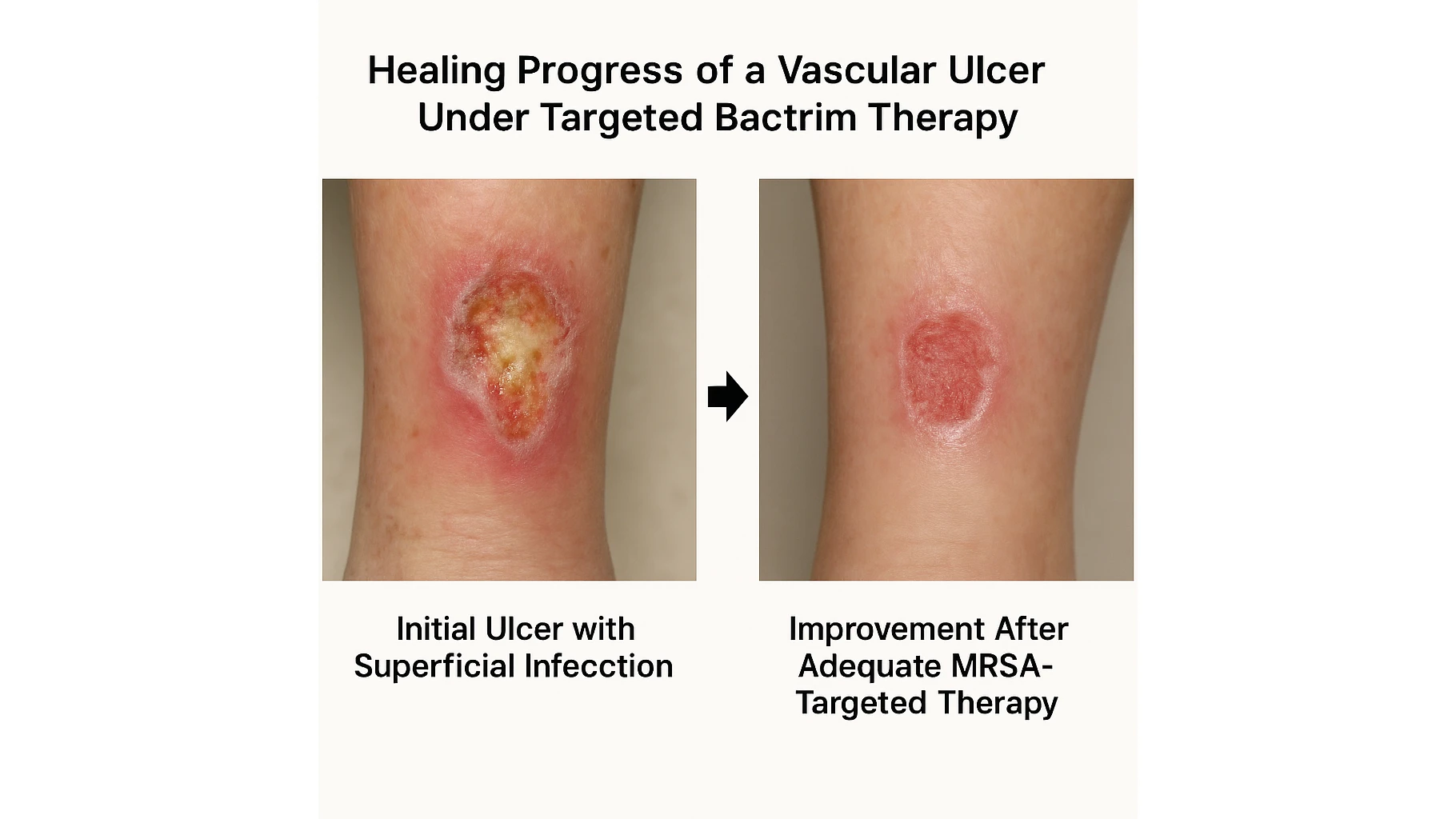
When Bactrim Should NOT Be Used
Although Bactrim is effective for many limb infections in vascular patients, there are clear situations where it is unsuitable or even potentially harmful. Chronic ischemic ulcers often progress from superficial colonization to deeper tissue involvement, introducing anaerobes, mixed Gram-negative species, and organisms residing in necrotic tissue. Because Bactrim does not provide anaerobic coverage, it cannot be used as monotherapy for infections showing necrosis, malodor, fluctuance, or crepitus.
Patients with severe diabetic foot infections, deep space involvement, or suspected osteomyelitis require broader-spectrum agents or combination therapy, often delivered intravenously. Bactrim should also be avoided in patients with significant renal impairment unless dosing is carefully adjusted, as its components accumulate and may increase toxicity risks. Allergy to sulfonamides remains an absolute contraindication, and clinicians should screen carefully for prior hypersensitivity reactions.
Another important consideration is drug interactions. Bactrim increases serum potassium and potentiates the anticoagulant effect of warfarin two issues particularly relevant in vascular patients who may already be taking ACE inhibitors, spironolactone, or anticoagulant therapy. Monitoring potassium and INR is essential to avoid life-threatening complications.
These limitations underscore the importance of selecting therapy based not only on antimicrobial spectrum but also on patient-specific risks. This principle aligns with the antibiotic safety considerations discussed in article Ciprofloxacin in Surgery, where the complexities of fluoroquinolone use illustrate why matching pharmacologic properties to patient physiology is essential in vascular care.
Comparative Effectiveness: Bactrim vs Ciprofloxacin, Keflex, and Amoxicillin
Antibiotic choice in vascular limb infections must account for perfusion deficits, biofilm formation, and the high likelihood of resistant organisms. For this reason, comparing Bactrim with other commonly used agents helps establish where it fits into modern treatment pathways.
Bactrim vs Ciprofloxacin
Ciprofloxacin is effective against many Gram-negative organisms but offers unreliable coverage against MRSA the most frequent pathogen in early ischemic ulcer infections. Furthermore, in vascular patients, the risk of tendinopathy and tendon rupture associated with fluoroquinolones becomes more clinically significant.
Bactrim vs Keflex (cephalexin)
Keflex remains a classical agent for uncomplicated cellulitis but is ineffective for MRSA and many Gram-negative strains found in chronic wounds. In patients with compromised perfusion, inadequate coverage contributes to treatment failure. For this reason, Keflex may be inappropriate for infections involving staphylococcal resistance or mixed flora that require broader action.
Bactrim vs Amoxicillin or Amoxicillin–clavulanate
While amoxicillin–clavulanate provides anaerobic and mixed-flora coverage, it often lacks reliability against MRSA. In early ischemic ulcers dominated by staphylococcal species, Bactrim delivers more precise coverage. amoxicillin alone has even narrower limitations and is rarely appropriate in vascular-limb infections.
This comparative framework clarifies why Bactrim has become a frontline oral therapy for MRSA-positive or suspected staphylococcal infections in vascular patients offering strong coverage without the systemic risks associated with broader-spectrum agents.
Table: Bactrim Indications and MRSA Coverage (2025 Update)
Clinical Utility Table
| Clinical Situation | MRSA Risk | Suitable for Bactrim? | Notes |
|---|---|---|---|
| Non-necrotic trophic ulcer | Medium | Yes | Good soft-tissue penetration; early intervention beneficial |
| Superficial infection in diabetic patient | High | Yes | Effective against MRSA; outpatient-friendly |
| Deep or necrotic ulcer with odor | High | No | Requires anaerobic + broad IV coverage |
| Postoperative wound after vascular bypass | Medium | Yes / Selective | Consider Gram-negative flora and perfusion status |
This table reflects modern 2025 usage trends, where Bactrim is favored for early, localized soft-tissue infections but excluded from cases that involve deeper or anaerobic pathology.
Dosing Strategies and Duration for Vascular Patients
Standard adult dosing for Bactrim DS is one tablet twice daily, providing adequate tissue penetration for managing early or superficial limb infections. In MRSA-positive wounds, courses typically last 7–14 days, with duration depending on wound response, perfusion quality, and comorbidities.
Because vascular patients often have complex treatment profiles, clinicians should consider the following factors when selecting a dosing plan:
- Assess renal function before initiation; reduce dose if creatinine clearance is low.
- Monitor serum potassium, especially in patients taking ACE inhibitors, ARBs, or potassium-sparing diuretics.
- Evaluate for potential drug interactions (e.g., warfarin, spironolactone).
- Reassess the wound clinically after 48–72 hours to confirm treatment response.
- Consider intravenous or broader-spectrum therapy if there is no improvement.
Elderly patients or those receiving multiple cardiovascular medications may require individualized dosing adjustments and closer laboratory monitoring.
Safety, Contraindications, and Monitoring
Bactrim is generally well tolerated, but vascular patients represent a higher-risk group due to frequent comorbidities such as chronic kidney disease, diabetes, hypertension, and polypharmacy. These conditions increase the likelihood of adverse events or drug interactions, making careful monitoring essential.
The most clinically important risks include:
- Hyperkalemia, especially when combined with ACE inhibitors, ARBs, or spironolactone
- Renal function decline due to accumulation of trimethoprim–sulfamethoxazole
- Warfarin potentiation, leading to elevated INR and bleeding risk
- Hypersensitivity reactions, particularly in patients with sulfonamide allergy
- Gastrointestinal side effects, such as nausea or mild abdominal discomfort
In vascular patients, electrolyte and renal monitoring should occur both at baseline and during treatment.Early recognition of laboratory abnormalities prevents avoidable complications and allows clinicians to tailor therapy without compromising efficacy. For most patients, Bactrim remains safe when properly monitored and used within established dosing guidelines.
FAQ – Practical Questions for 2025
Is Bactrim reliable for MRSA in vascular patients?
Yes. Bactrim remains one of the most effective oral options for MRSA in patients with limb ischemia or chronic ulcers.
Can Bactrim treat deep infections or necrotic wounds?
No. Deep or necrotic infections require broader-spectrum therapy, often including anaerobic coverage and intravenous treatment.
How long should Bactrim be used for a limb infection?
Most MRSA-soft tissue infections respond within 7–14 days, depending on vascular status and wound response.
What laboratory tests are required during treatment?
Serum potassium, creatinine, and in patients on warfarin INR monitoring are essential.
Can Bactrim be used after vascular surgery?
Yes, selectively. It is appropriate for superficial postoperative infections, but broader therapy may be needed if deeper contamination or graft involvement is suspected.





























